Generated Narrative: StructureDefinition MedicinalProductLM
| Name | Flags | Card. | Type | Description & Constraints |
|---|---|---|---|---|
 | 0..* | Base | Medicinal product (model) Instances of this logical model can be the target of a Reference | |
 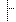 | 0..* | CD, II | Identifier or product code for the product, independently of the level of product. If several identifiers are specified, they shall not have conflicting meanings or very different granularities. | |
 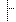 | 0..* | CD | Classification (e.g. ATC; narcotic/psychotropic; orphan drug; etc.) | |
 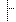 | 0..1 | ST | Name of the product (full name, invented name, other). When the product has different names, the appropriate one for the context should be used. Translations of names can be provided. | |
 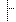 | 0..1 | Class | Marketing authorisation holder of manufacturer of the medicinal product. Relevant for identifying the exact product. | |
   | 0..1 | ST | Name of the organisation holding the authorisation for marketing/mahufacturing | |
   | 0..* | II | Identifier of the organisation and/or its physical location | |
  | 0..1 | CD | Dose form(s) on a product level. Dose form for a single package item is defined below. | |
  | 0..* | PQ | Overall amount of product in one package (100ml; 20 tablets; 1 creme & 6 pessaries) | |
  | 0..* | Class | A medication item. For combination packs, this can be manufactured items with each item having its own dose form and ingredients+strengths defined | |
   | 0..1 | CD | Dose form | |
   | 1..* | Class | Ingredients | |
    | 0..1 | CD | Role (active ingredient, excipient). Typically excipients are not needed, so by default active ingredients are expected. | |
    | 1..1 | CD | Substance | |
    | 0..* | Class | Concentration or presentation strength | |
     | 1..1 | RTO | Concentration or presentation strength of the precise active ingredient | |
     | 0..1 | CD | Type of strength that is expressed | |
     | 0..1 | CD | Substance that the strength refers to, if different from the main substance | |
   | 0..1 | CD | Unit of presentation for the manufactured item (tablet, vial, tube). Typically, the smallest countable object in the package. | |
   | 0..1 | PQ | Manufactured item quantity for liquids (3ml/vial) | |
   | 0..1 | PQ | Number of such manufactured items in this product (5 vials) | |
  | 0..* | Class | Administration device included in the product | |
   | 1..1 | PQ | Number of such devices | |
   | 1..1 | CD | Device coded | |
  | 0..* | Class | Other features of the product | |
   | 1..1 | CD | A code expressing the type of characteristic | |
   | 0..1 | BL, CD, ST, PQ, DT, INT, REAL, RTO | Description of the characteristic | |
  | 0..1 | Class | Additional information about product - for example, a reference to a product catalogue | |